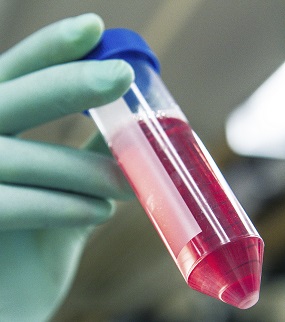
Suspending and Homogenizing Cell Pellets
When working with biological cell cultures, homogenizing cell pellets can have multiple purposes. Two popular examples include preparation for protein extraction or creating an inoculum. In protein extraction, cells need to be separated from each other to ensure effective contact with the protease inhibitor cocktail, the lysis mechanism and the extraction buffer. For inoculation purposes, it is key to have homogeneous distribution so the cell number in the volume of inoculum is consistent. For best results in either of these procedures, it is critical to create a uniform distribution of cells in the suspension medium.

The cells are first separated from old and consumed media. Centrifugation drives the cells to the bottom of the vessel, resulting in a compacted mass so the used media can be removed by decanting. Washing buffer or fresh culture media is added, then the cell pellet is agitated, dislodging the pellet from the bottom of the vessel. Continued agitation separates the cells by breaking the compacted pellet into small clumps, then to single cells, resulting in a homogenized cell suspension. Washing and spinning can be repeated as needed, usually keeping the cells on ice during the process.
There are several considerations regarding the best method for suspending cells. Many cell types are sensitive to the mechanical stress forces created by vortexing. Pipetting can be an extended manual process for larger pellets in small suspension volumes. The scale of the cell culture could create suspensions too viscous for magnetic stirring, or may require an additional step of transferring the pellet and medium to a vessel able to accommodate the stir bar.
Caframo’s BDC2002 overhead stirrer can be an effective option to address many of these issues. Impeller selection and speeds from 40-2002 rpm can be matched to create balanced agitation that minimizes damage and successfully separates cells. The adjustable chuck allows easy changes to the correct impeller size to fit the vessel being used, and the through shaft provides the ability to quickly raise the impeller out of the way when the pellet is homogenized. Direct illumination on the vessel is provided by the Stirlight™ feature. With the vessel nestled in ice, users have their hands free to prepare for subsequent steps once the desired homogenized cell suspension is ready. Contact us to learn about the options available.
|

|
|
References:
Cho D, Collins MT. (2006). Comparison of the Proteosomes and Antigenicities of Secreted and Cellular Proteins Produced by Mycobacterium paratuberculosis. Clinical and Vaccine Immunology, 13(10), 1155-1161. doi:10.1128/CVI.00058-06
Sung N, Collins MT. (1998) Thermal Tolerance of Mycobacterium paratuberculosis. Applied and Environmental Microbiology, 64(3), 999-1005.
GE Healthcare Life Sciences. (2010). Sample collection, stabilization, and protein extraction. In:. Protein Sample Preparation Handbook. Retrieved from: http://www.gelifesciences.com/webapp/wcs/stores/servlet/catalog/en/GELifeSciences-us/service-and-support/handbooks/

