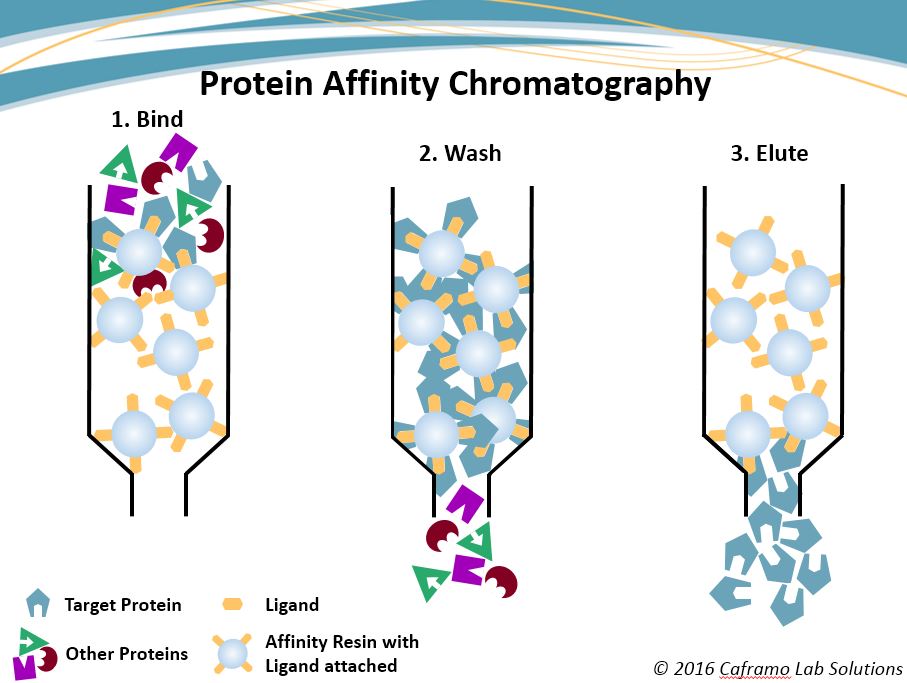
Protein Affinity Chromatography
In general, chromatography occurs with a solid support (often called a resin or matrix) packed into a column, forming a stationary phase. The mobile phase, a solution containing a mixture of molecules, is moved through the column from one end to the other. The chemical and physical differences in the molecules leads to different rates of passage through the column matrix, leading to separation.
Affinity chromatography is a powerful version of chromatography used to separate and purify molecules of interest, particularly biological macromolecules such as proteins. The high selectivity and resolution of this technique make it popular for both laboratory and process-scale applications.
Affinity chromatography has high selectivity and resolution for purifying molecules of interest.
How affinity chromatography works is often explained using a lock and key analogy, and is illustrated in the graphic below. A specific ligand, the key, is coupled to the resin. The mobile phase, a complex mixture of many proteins and other cellular debris, is brought into contact with the prepared resin. Within the mobile phase is the lock, the target protein whose chemical properties create a uniquely strong affinity to the key. During binding, the “key” ligands catch and hold the corresponding “lock” proteins as the protein mixture comes into contact with the ligands, holding them to the resin. Washing follows where the other proteins and contaminants, not being fixed to the resin, are washed away leaving only the protein of interest behind. Elution, the final step, now releases the purified protein from the resin so it can be collected for further purification or other processing.
Overhead stirrers are especially beneficial in several steps of the affinity chromatography process:
- Coupling ligands to the support matrix
- Equilibrating a slurry of matrix beads in binding buffer
- Suspending a matrix slurry in preparation to pack column
- Mixing resin with large volumes (> 200 mL) cell lysate during binding phase
- Regenerating used resin
- Suspending resin as first step for removing fines
Many affinity resin and column product inserts specifically warn against using a magnetic stirrer
Many affinity resin and column product inserts specifically warn against using a magnetic stirrer, recommending an overhead or paddle stirrer for the steps listed above. The critical factor is avoiding high shear stress on the resin, yet have successful homogenous mixing. Stirring with a magnetic stir bar risks abrading or shearing the beads, resulting in undesired “fines” – a chromatography term for fine fragments. Overhead stirring must be low shear and the minimal amount possible, to avoid overmixing. Unfavourable consequences of fines include decreased and non-uniform flow through the column, detrimental to separation resolution and consistency. Degradation to the particles can also result in reduced resin lifetimes.
Caframo overhead stirrers provide features and flexibility for low shear suspending that is consistent for repeatable results. The digital display on all models shows speed in rpm, which can be gradually adjusted in 1 rpm increments with a touch of a button. The 2010 includes a timer which can stop stirring to avoid overmixing the resin. The chuck and through shaft allow easy installation and positioning of low shear impellers to have sufficient agitation to suspend the matrix and minimize risk of fines. Contact us to find the best solution for your needs.

|
|

|
References:
Applied Biosystems (n.d.) Poros® HP Glass Columns for Preparative Chromatography – Column Packing Instructions [Product Information] Retrieved August 2016, from http://tools.thermofisher.com/content/sfs/manuals/cms_041701.pdf
BioLegend (n.d.) Anti-c-Myc Tag (9E10) Affinity Gel [Technical Bulletin] Retrieved August 2016, from https://www.biolegend.com/media_assets/support_protocol/Anti-c-Myc_Agarose_Technical_121313.pdf
Bemberis I, Noyes A, and Natarajan V. (2003, July) Column Packing for Process-Scale Chromatography: Guidelines for Reproducibility. In BioPharm International Guide, 23-30. Retrieved August 2016, from http://www.academia.edu/22088554/BioPharm_International_Guide_Column_Packing_for_Process-Scale_Chromatography_Guidelines_for_Reproducibility
Bio-Rad (n.d.) Introduction to Affinity Chromatography. Retrieved August 2016, from http://www.bio-rad.com/en-us/applications-technologies/introduction-affinity-chromatography
Biotech Support Group (n.d.) NuGel™ Poly-Aldehyde – Polymer Coated Silica Affinity Matrices. [Product Information] Retrieved August 2016, from http://www.biotechsupportgroup.com/v/vspfiles/templates/257/pdf/NuGel%20Affinity%20Matrices %20Product%20Sheet%20PolyAldehyde%20073015MK.pdf
Constans, A. (2005, Feb) Tag! Purifying Proteins with Affinity Chromatography. The Scientist. Retrieved August 2016, from http://www.the-scientist.com/?articles.view/articleNo/16257/title/Tag–Purifying-Proteins-with-Affinity-Chromatography/
Deppert, W.R., & Lukačin R. (2000) Hydroxyapatite Chromatography. In Kastner, M. (Ed.), Journal of Chromatography Library Volume 61: Protein Liquid Chromatography (pp. 271-300). Amsterdam, Netherlands: Elsevier Science B.V.
GE Healthcare Bio-Sciences AB (2014) Affinity Chromatography Columns and Media Selection Guide. Retrieved August 2016, from https://www.gelifesciences.com/gehcls_images/GELS/Related%20Content/Files/1461771565566/ litdoc18112186_20160427173857.pdf
ThermoFisher Scientific (n.d.) Overview of Affinity Purification. Retrieved August 2016, from https://www.thermofisher.com/ca/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-affinity-purification.html
Toumi, A., Dingenen, J., Genolet, J., Ludemann-Hombourger, O., Kiesewetter, A., Krahe, M., Morelli, M., Schmidt-Traub, H., Stein, A., & Valery, E. (2012) Chromatography Equipment: Engineering and Operation. In Schmidt-Traub, H., Schulte, M., & Seidel-Morgenstern, A. (Eds.), Preparative Chromatography (2nd ed.) (pp. 199-271). Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA

